Wonderful Info About How To Prevent Corrosion On Iron Nails

When the water is boiling vigorously,.
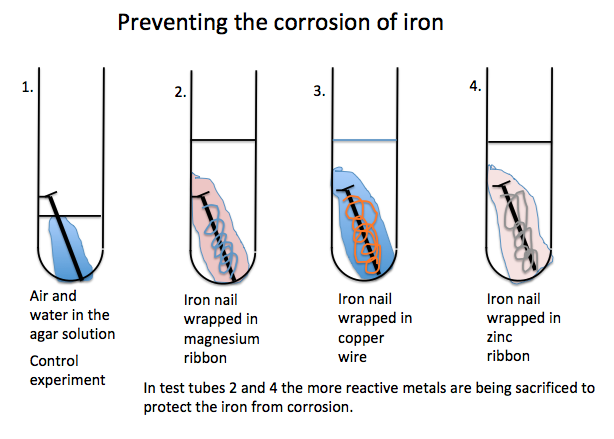
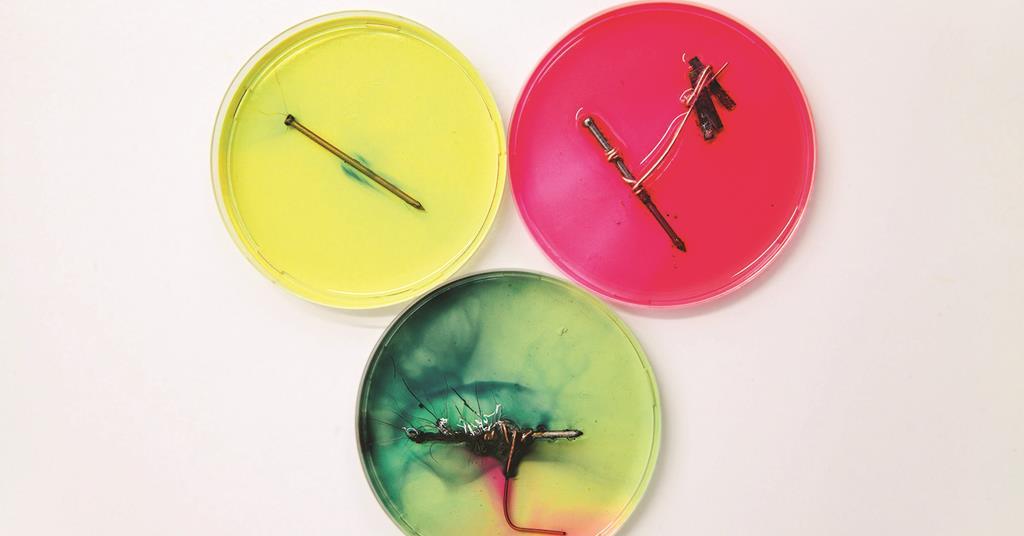
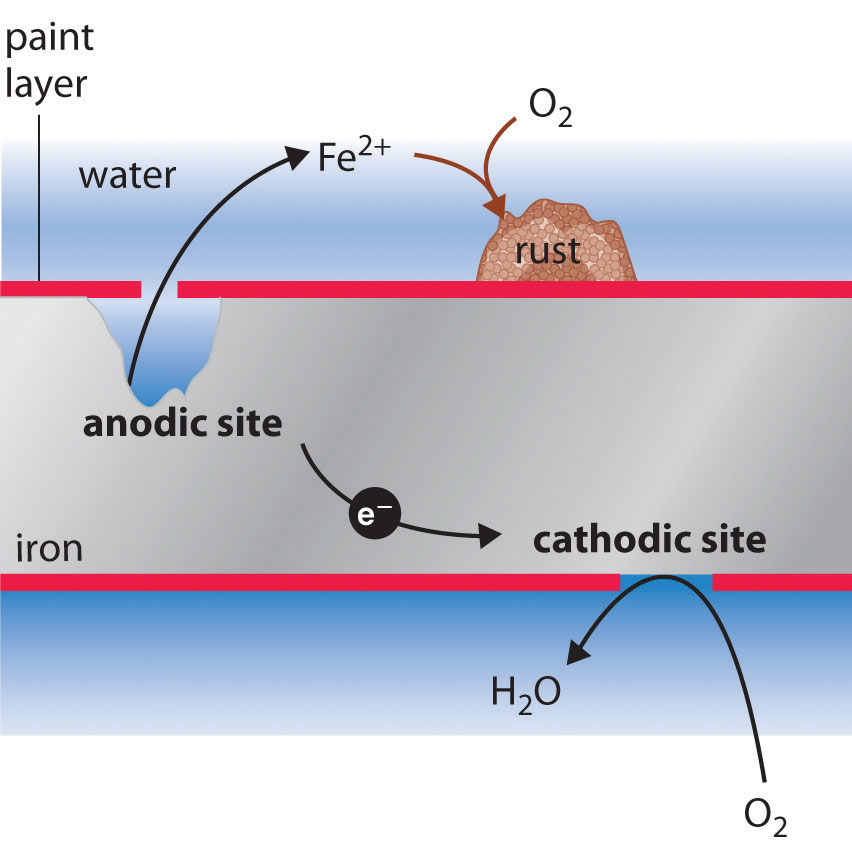
How to prevent corrosion on iron nails. Metals situated above iron (fe) on the. Corrosion of an iron nail in water. When iron is placed in contact with a more active metal (one that is more easily oxidized), the more active metal will be corroded instead of the iron.
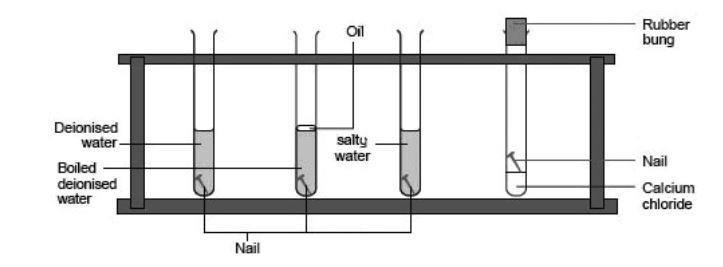
Metals high in the series can be used to “protect” less reactive metals (say, because they are. Oxygen can be excluded by storing the. Using the balance, measure out 2.00 g of agar powder.
Corrosion of an iron nail in water. Posted by 1 year ago. Connect the first nail to the less reactive metal copper cu, leave the second one as is, and connect the third one to.
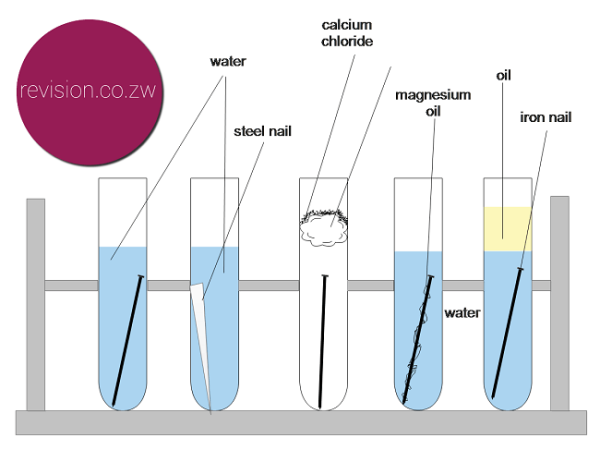
Galvanizing coats iron or steel in zinc to protect from rust. •hms samarang •able to prevent corrosion of the iron nails by using zinc or iron ‘protectors’ •effective at preventing the corrosion of the iron, but also stops the copper from corroding •the. We would like to show you a description here but the site won’t allow us.
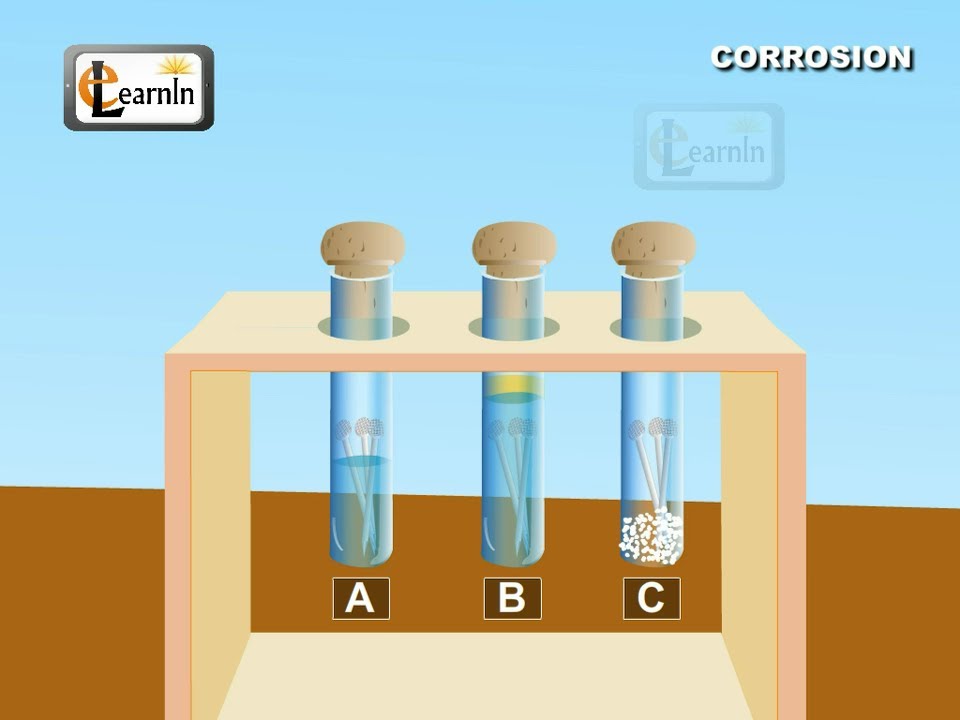
The higher the metal is in the series, the more reactive it is. Corrosion of iron continued 4 216 inn cientific inc ihts esered teacher’s notes for guided inquiry corrosion of iron demonstration procedure 1. Clean two iron nails with sandpaper or steel.
Use of desiccant drying agents in this storage are also helpful. Rusting of iron can be prevented by applying grease or oil: An understanding of the activity series investigated in experiment 6 suggests that one way of preventing the corrosion of iron is to protect it with a more active metal.